Novo Nordisk has dropped its patent infringement lawsuit against Hims & Hers Health after the pair struck an agreement allowing Hims to distribute branded Ozempic and Wegovy - including the new oral Wegovy pill - through its platform at the same price as other telehealth providers.
"We have decided to drop the current court proceedings," Novo CEO Mike Doustdar told CNBC, adding that the company reserves the right to refile "if needed".
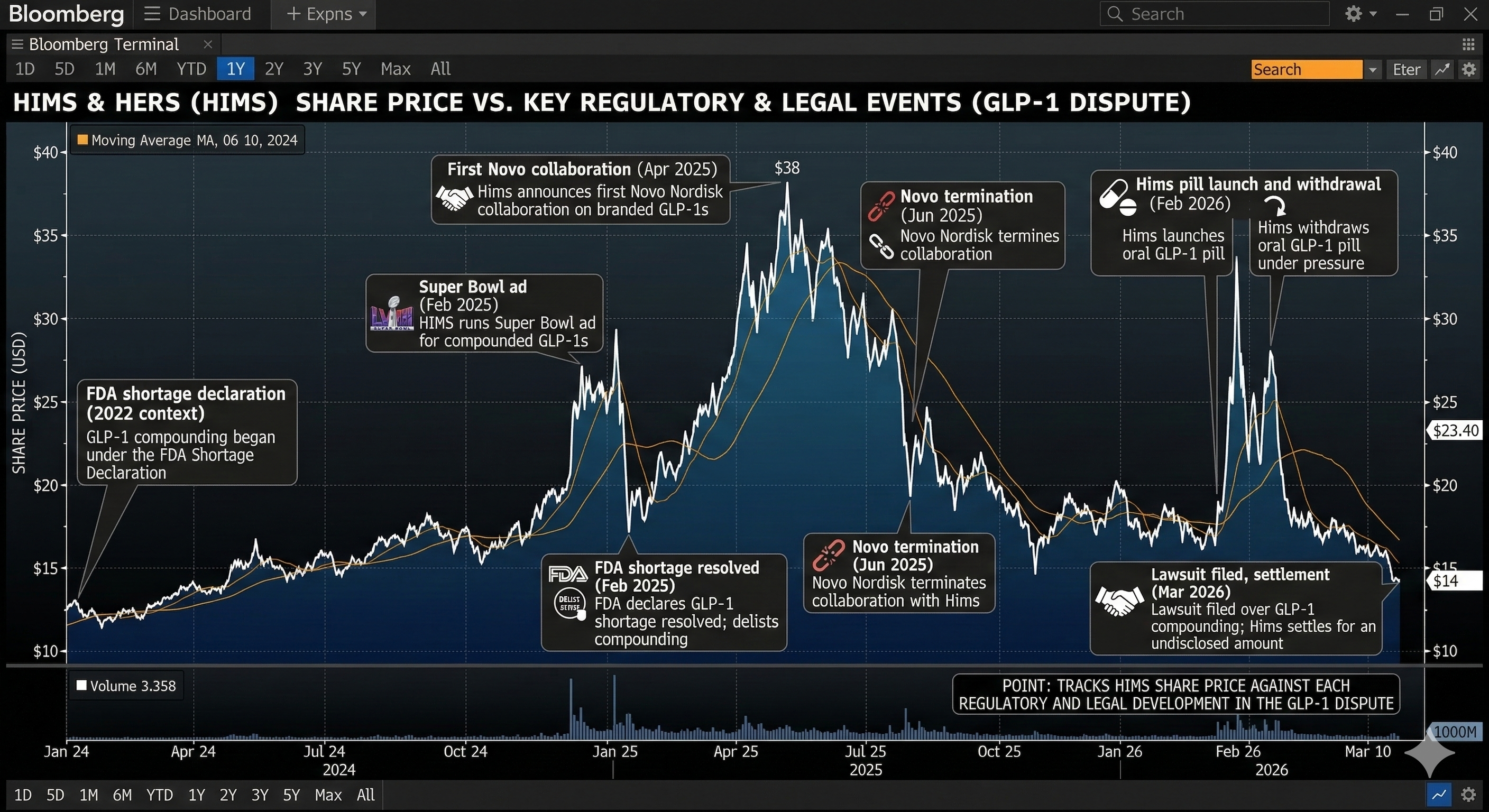
Shares of Hims & Hers jumped more than 36% in Monday morning trading, though they remain well off their 52-week high of around $70. United States-listed shares of Novo Nordisk rose 1.8%.
How the loophole opened - and closed
The dispute has its roots in 2022, when the FDA placed semaglutide on its official drug shortage list. That listing authorised compounding pharmacies to obtain the active ingredient and produce their own versions to meet patient demand.
Hims & Hers began selling compounded injectable semaglutide in May 2024 under that provision.
On 9 February last year, the company ran a Super Bowl commercial called "Sick of the System", which reached an estimated 127 million viewers and promoted its compounded formulation as a lower-cost alternative to branded GLP-1 drugs.
Then on 21 February, the FDA declared the semaglutide shortage resolved for all presentations of the drug. Hims shares fell a sharp 25% on the news.
In April 2025, Novo and Hims announced a collaboration to expand access to obesity treatment. Novo terminated the arrangement in June 2025, accusing Hims of mass-compounding its drugs under the guise of personalisation and using deceptive marketing.
On 5 February 2026, Hims launched a compounded oral semaglutide pill priced at $49 a month - against Novo's branded Wegovy pill at $149 a month on its NovoCare platform.
The FDA responded within 24 hours, announcing it would take "decisive steps" to restrict mass marketing of non-approved GLP-1 products. The Department of Health and Human Services referred Hims to the Justice Department for potential violations of the Federal Food, Drug and Cosmetics Act.
Two days after launch, Hims withdrew the product. Novo filed its patent infringement suit the following Monday, describing the pill as "an unapproved, inauthentic and untested knockoff" of semaglutide.
Terms of the settlement
Under Monday's agreement, Hims will stop advertising compounded GLP-1 drugs, reserving compounded versions for cases where they are medically necessary.
FDA Commissioner Marty Makary said he welcomed the deal, noting Hims would keep branded drug prices unchanged and limit compounding to FDA-compliant cases.
Hims reported fourth-quarter revenue of $617.82 million, with full-year 2026 guidance of $2.7 billion to $2.9 billion and adjusted EBITDA of $300 million to $375 million.
Meanwhile, Novo Nordisk said it had issued more than 600,000 Wegovy pill prescriptions as at the time of the announcement.